 During method development and its application, we found that hydrolysis of the succinimide ring of the linker was ubiquitous, taking place at many stages during the lifetime of the PDC including in the initial drug product, in vivo in circulation in the animals, and ex vivo during the trypsin digestion step of the sample preparation. The tryptic peptide containing the linker attached to the payload was used as a surrogate of the conjugate and monitored by LC/MS/MS analysis. The His-tagged proteins were isolated from biological samples using immobilized metal affinity chromatography (IMAC), followed by trypsin digestion. In our current work for a protein-drug conjugate (PDC) using the Centyrin scaffold, a similar concept to ADCs but with smaller protein size, an alternative method to quantify the conjugate by using a surrogate peptide approach, was utilized. Existing approaches to quantify the conjugated species of ADCs involve a ligand binding assay (LBA) for conjugated antibody or hybrid LBA/liquid chromatography/tandem mass spectrometry (LC/MS/MS) for quantitation of conjugated drug. Among them, conjugate is the most challenging to measure, because it requires detection of both small and large molecules as one entity. 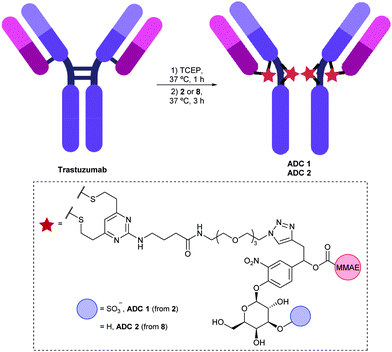
To fully describe the pharmacokinetics (PK) of an ADC, several analytes are commonly quantified, including total antibody, conjugate, and payload. Bioanalysis of antibody-drug conjugates (ADCs) is challenging due to the complex, heterogeneous nature of their structures and their complicated catabolism. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
